Kinases are a family of enzymes that catalyze the transfer of a phosphate moiety from a high-energy, phosphate-donating molecule, such as ATP, to its specific substrate. This process is referred to as phosphorylation, where the high-energy ATP molecule donates a phosphate group to the substrate, producing a phosphorylated substrate and ADP (Figure 1). Because of the high levels of energy released during the breakage of phosphoanhydride bonds, kinases are necessary to stablize this reaction
Kinases

Figure 1. Protein kinase reaction scheme.
Kinases are part of the larger family of phosphotransferases, which are classified into groups by the substrate they act upon such as protein kinases, lipid kinases or carbohydrate kinases. The phosphorylation state of a molecule can affect its activity, reactivity, and its ability to bind to other molecules. Kinases are particularly prominent in various cellular pathways, including metabolism, cell signaling, protein regulation, and cellular transport, which makes them one of the most important target classes in drug discovery for the development of novel therapeutic drug candidates or kinase inhibitors.
Protein Kinases
Protein kinases play a key role in cellular activation processes. By adding phosphate groups to substrate proteins, protein kinases direct the activity, localization and function of many proteins, and serve to regulate the activity of vital cellular processes. They constitute one of the largest and most functionally diverse gene families with more than five hundred different kinases have been identified in humans.
Types of Human Protein Kinases:
- AGC kinases: containing PKA, PKC and PKG.
- CaM kinases: containing the calcium/calmodulin-dependent protein kinases.
- CK1: containing the casein kinase 1 group.
- CMGC: containing CDK, MAPK, GSK3 and CLK kinases.
- STE: containing the homologs of yeast Sterile 7, Sterile 11, and Sterile 20 kinases.
- TK: containing the tyrosine kinases.
- TKL: containing the tyrosine-kinase like group of kinases.
Developing kinase assays requires methods that detect the conversion of a substrate by indentifying either the formation of a phosphopeptide, the disappearance of ATP, or the formation of ADP. Most commercially available protein kinase assay kits are designed to monitor either phosphopeptide formation or ATP depletion. However, most phosphopeptide formation-based assays are dependent on antibodies or have special requirements for the peptide substrate, while ATP depletion assays can encounter a range of interferences due to the inhibition or activation of luciferase by various biological compounds. At AAT Bioquest our ATP and ADP detection kits do not use antibodies or special substrates, experience minimal interference from biological compounds, and can be used for monitoring a kinase activity generically.
Amplite™ Universal Fluorimetric Kinase Assay
Amplite™ Universal Fluorimetric Kinase Assay Kit (Cat No. 31001) is a convenient non-radioactive and no-wash homogeneous assay for measuring kinase activity. This kit utilizes an enzyme-coupled reaction to monitor the formation of ADP, which is directly proportional to phosphotransferase activity. The characteristics of its high sensitivity (<0.2 µM ADP), broad ATP tolerance (1-300 µM) and non-antibody based method make it an ideal kit for determining kinase Michaelis-Menten kinetics and for screening and identifying kinase inhibitors. The assay can be performed in a convenient 96-well or 384-well microtiter-plate formate and easily adapted to automation with no separation steps required.
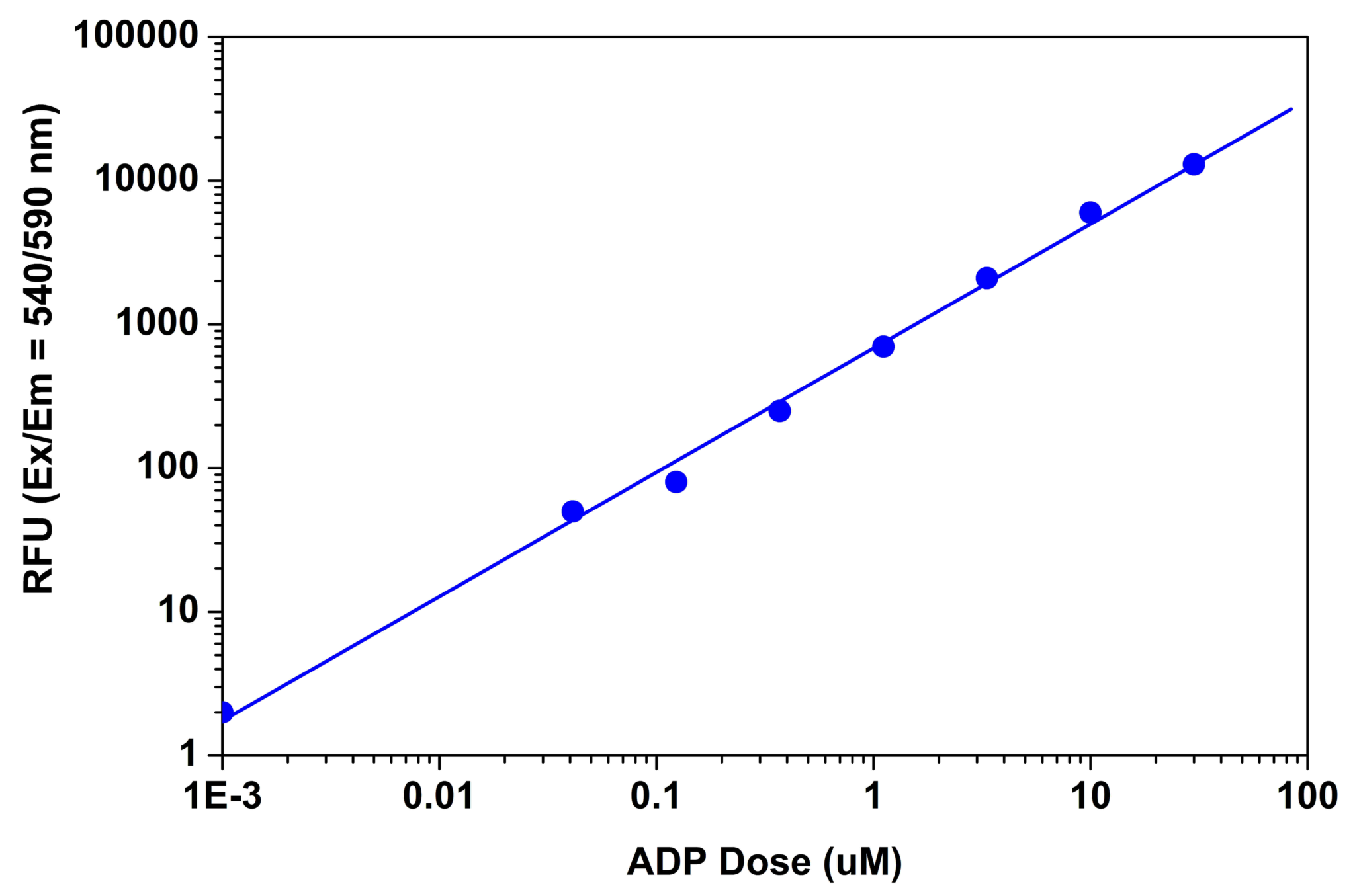
Figure 2. ADP dose response was measured with Amplite™ Universal Fluorimetric Kinase Activity Kit in a solid black 384-well plate using a Gemini fluorescence microplate reader (Molecular Devices).
Enterokinase
Enterokinase (also called enteropeptidase) is a serine protease produced by cells in the duodenal wall. As a key enzyme in the digestion system of humans and other animals, enterokinase converts trypsinogen (an inactive substance secreted by the pancrease) into its active form trypsin. Trypsin, which catalyzes the hydrolysis of peptide bonds, breaks down proteins into smaller peptides. These peptide products are subsequently hydrolyzed into amino acids via other proteases faciliting their absorption into the blood stream. Recent evidence suggests that the inhibition of enterokinase activity may cause anti-tumor effects through suppressing proteases involved in carcinogenesis and metastasis. Therefore, highly selective and sensitive detection of enterokinase plays a key role in biochemical applications.
Amplite™ Colorimetric Enterokinase Activity Assay
Amplite™ Colorimetric Enterokinase Activity Assay Kit (Cat No. 11410) offers a sensitive assay for quantifying enterokinase activity. After cleavage of enterokinase, the enterokinase substrate can be detected by EK Yellow™ using an absorbance microplate reader at 405 nm. With the Amplite™ Colorimetric Enterokinase Activity Assay Kit, as low as 3 ng/mL enterokinase can be detected in a 100 µl reaction volume.
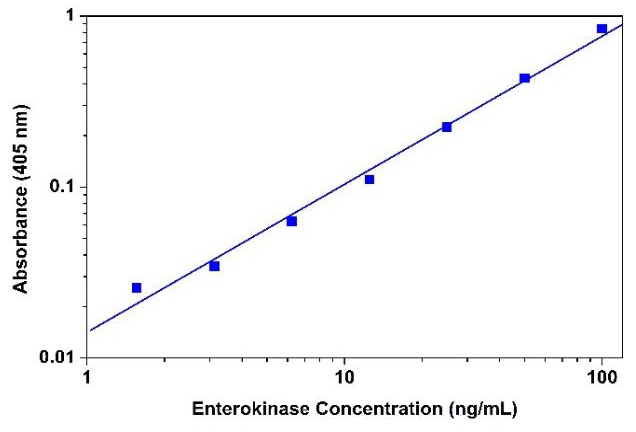
Figure 2. Enterokinase dose response was measured with Amplite™ Colorimetric Enterokinase Activity Assay Kit in a 96-well clear bottom microplate using a SpectraMax microplate reader (Molecular Devices) with path check on mode.
Amplite™ Universal Fluorimetric Kinase Assay Kit
Most of commercial protein kinase assay kits are either based on monitoring of phosphopeptide formation or ATP deletion. For the kinase assay kits that are based on detection of phosphopeptides one has to spend time and efforts to identify an optimized peptide…
| Catalog | Unit Size | Additional Information |
| 31001 | 250 Tests | Red Fluorescence |
| 31002 | 500 Tests | Red Fluorescence |
Amplite™ Colorimetric Enterokinase Activity Assay Kit
Enterokinase (also called enteropeptidase) is a serine protease produced by cells in the duodenal wall and is a key enzyme in human and animal digestion system. Enterokinase converts trypsinogen into its active form trypsin, resulting in the subsequent activation…
| Catalog | Unit Size | Additional Information |
| 11410 | 200 Tests | N/A |