Polylysine (Fig. 1) is a bio-compatible cationic polymer. It comes in both L or D type, with the former digestible by some enzymes and latter more stable. Under physiological conditions, the amine group on the lysine molecules are protonated. The cationic nature makes it an attractive molecular coating for the adhesion of negatively charged biomolecules, particularly DNAs. It is also ideally suited for the adhesion of cells, since the cell surface is known to be negatively charged. Thus, polylysine coatings are widely used for the adhesion of cells in cell biology experiments, for the fabrication of DNA or cell microarrays, and as biocompatible coatings for biomaterials, as illustrated in Fig. 2. In addition, poly-lysine coatings are also used as anchors for other biocompatible polymers, such is the case of polylysine-polyethyleneglycol (PEG) block copolymers.
The process of forming a polylysine coating in principle very simple. For a common substrate such as glass, the clean surface usually possesses net negative charge. As a result, polylysine can easily adsorbed onto the negatively charged surface. For sufficiently high polylysine molecular weight (>100 kD) and sufficient electrostatic attraction, the adsorbed polylysine layer can be robust for subsequent use in a variety of experiments. However, the seeming simple adsorption process is not easily reproducible. The amount of polylysine adsorption and the stability of the adsorbed layer can be low. As a result, one often obtains a polylysine coating with very low surface coverage (average thickness ~ 1 nm). This introduces uncertainty and irreproducibility for subsequent adsorption of DNAs or cells. In addition, it can lead to substantial wasting of the very expensive polylysine molecules that are washed off
during coating and during subsequent exposure to solutions.
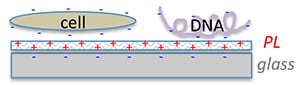 Fig. 2. Schematic illustration of the adsorption of cell or
Fig. 2. Schematic illustration of the adsorption of cell or
DNA molecule (with negative charges) on the positively
charged polylysine coated glass surface.
Taking advantage of our extensive knowledge and expertise in surface coating chemistry, as well as quantitative surface characterization techniques, e.g., X-ray photoelectron spectroscopy (XPS), ellipsometry, atomic force microscopy (AFM), and attenuated total internal reflection Fourier transform infrared (ATR-FTIR) spectroscopy, we have optimized the fabrication of polylysine coatings on glass, quartz, and other hydrophilic surfaces. Our polylysine coatings are homogeneous, stable, and reproducible, and present a high density of positive charge under physiological conditions. You can have peace of mind when you apply our high-density polylysine coating products in your research and development projects. Talk to our scientists about your needs and we will help you achieve your goals.
Copy Right© 2015 Athena Guo. All rights reserved.