Useful Terminology
Agglomeration and Aggregation
Aggregation and agglomeration are terms that are widely used by nanotechnology researchers to refer to nanoparticles that have associated into a cluster composed of two or more nanoparticles. Use of aggregate vs. agglomerate is industry specific, but in the nanoparticle field the two terms are equivalent and can be used interchangeably. Regardless of which term you choose, the agglomeration state of a nanoparticle is a critical parameter that should be known at each stage of nanomaterial processing. The vast majority of dried nanoparticles are permanently agglomerated into clusters that consists of tens, hundreds, or even thousands of individual nanoparticles, significantly increasing the effective size of the nanoparticles and potentially altering the nanoparticle’s physical and optical properties. The figure to the right shows a schematic of two thousand 10 nm nanoparticles dispersed individually (Top) or clustered into 100 nm agglomerates (Bottom). It is clear that the properties of these two nanomaterials are very different even though each contains the same number of identically sized nanoparticles.
 Applying shear forces to nanoparticle agglomerates can break the agglomerates into two or more pieces. However, even with the most powerful dispersion mechanisms (probe sonication, microfluidization, milling), it is typically not possible to restore agglomerated particles back to a monodisperse suspension consisting only of individual nanoparticles. This is due in part to the relative strength of the van der Waals forces that bind particles together and the shear forces that can be applied on the nanoscale. In order to separate two agglomerated nanoparticles, forces in different directions have to be applied to each nanoparticle. At very small particle sizes it is simply not possible to generate sufficient microturbulence that creates a high enough force gradient to overcome the particle binding forces. Experimentally, the lower threshold for dispersed agglomerate size is ~200 nm for many different types of nanomaterials. For example, if 10 nm and 50 nm oxide nanoparticles are dried and extensively dispersed, the agglomerate size will be ~200 nm for both particle types. This is important because in many cases it is the agglomerate size not the primary particle size that is responsible for the nanoparticle’s diffusion and transport properties.
Applying shear forces to nanoparticle agglomerates can break the agglomerates into two or more pieces. However, even with the most powerful dispersion mechanisms (probe sonication, microfluidization, milling), it is typically not possible to restore agglomerated particles back to a monodisperse suspension consisting only of individual nanoparticles. This is due in part to the relative strength of the van der Waals forces that bind particles together and the shear forces that can be applied on the nanoscale. In order to separate two agglomerated nanoparticles, forces in different directions have to be applied to each nanoparticle. At very small particle sizes it is simply not possible to generate sufficient microturbulence that creates a high enough force gradient to overcome the particle binding forces. Experimentally, the lower threshold for dispersed agglomerate size is ~200 nm for many different types of nanomaterials. For example, if 10 nm and 50 nm oxide nanoparticles are dried and extensively dispersed, the agglomerate size will be ~200 nm for both particle types. This is important because in many cases it is the agglomerate size not the primary particle size that is responsible for the nanoparticle’s diffusion and transport properties.
The agglomeration state of solution-based samples can be measured using TEM analysis of dilute solutions where the number of nanoparticles in well separated clusters is counted. However, many cluster need to be counted and it is difficult to discriminate between pre-formed agglomerates and agglomerates that formed during the drying process. Alternatively, a Dynamic Light Scattering instrument can be used to measure the mean and distribution of hydrodynamic agglomerate size in solution. If the primary particle size has been previously determined with TEM, the average number of particles per aggregate can be determined. Another method is to use a Centrifugal Particle Sizer (CPS) which measures the time it takes for the nanoparticles to pass through a gradient. CPS measurments are preferred for multi-modal size distributions or for samples that have a high degree of polydispersity. The agglomeration state of dried samples can be measured by aerosolizing the powder and measuring the agglomerate size using an aerosol particle sizer (for sizes >500 nm) and a scanning mobility particle sizer (for sizes <500 nm).
Spectral Shifts: Blue-Shifting/Red-Shifting
For many types of nanoparticles, spectral peaks can shift due to changes in the size, shape, coating, or aggregation state of the nanoparticles. Blue-shifting refers to an electromagnetic response that is shifted towards shorter wavelengths (higher frequencies, higher energies). Red-shifting refers to a peak that shifts to longer wavelengths. For example, if the aspect ratio of a gold nanorod is decreased, the peak extinction wavelength blue shifts from 800 nm to 600 nm. Encapsulating a spherical silver nanoparticle with a silica shell will red shift the peak. Aggregation typically results in a red shift in the spectral characteristics of the nanoparticle suspension.
Extinction
Extinction refers to the sum of all light that is not transmitted through a sample. For nanoparticles, light can be removed by a scattering event or through absorption. Extinction is measured using a UV-Visible spectrophotometer. The proportion of extinction due to scattering or absorption changes dramatically for different sized nanoparticles. For example, 10 nm gold nanoparticles will be strongly absorbing while 100 nm gold nanoparticles will have an extinction that is dominated by scattering.
Extinction equations:
T = I/I0
Extinction = -log10T
T is the transmission, I is the intensity of the beam after passing through the sample, and I0 is the intensity of the incident source.
Grain Size/Primary Size
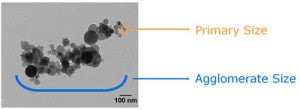
The grain size/primary size is the size of the smallest discrete element of an aggregate or agglomerate.
Most often, the primary size is measured by TEM imaging. Many nanomaterials consist of grains that are aggregated into larger agglomerates. It is important to distinguish between the grain/primary size and the agglomerate size.
Optical Cross-Sections
The optical cross section of a material is a measure of the intensity of the light that is absorbed or scattering by that material. When light hits the material, some of the photons are transmitted (pass through the target), while others are scattered or absorbed. The cross-section is a measure of the effectiveness with which photons are absorbed or scattered, with larger cross sections signifying that the material is increasingly effective. The sum of the absorption cross section and scattering cross section is referred to as the extinction cross section which is the total amount of light that is not transmitted through a sample. Another metric of interest is the extinction cross section divided by the geometric cross section. This value is the extinction efficiency and represents how strongly coupled a particle is to incident light. Surprisingly, for spherical silver nanoparticles, extinction efficiencies >20 can be obtained which is the highest efficiency obtainable for any material.

Optical Density
Optical density is a measure of the amount of light that is transmitted through a solution, filter, or sample. The optical density is the negative logarithm of the percentage of light transmitted through a solution: OD = -log10(T) where T is scaled to fall between 1 (100% transmission) and zero (0% transmission).
| Optical Density | % Transmission |
|---|---|
| 0 | 100 |
| 0.1 | 79.3 |
| 0.2 | 63.1 |
| 0.3 | 50.12 |
| 0.4 | 38.91 |
| 0.5 | 31.62 |
| 1 | 10 |
| 2 | 1 |
| 3 | 0.1 |
| 4 | 0.01 |
Optical Efficiency
The optical efficiency of a material is defined as the optical cross section divided by the geometric cross section. Reporting optical efficiency rather than optical cross section rescales the optical intensity by the nanoparticle size. Because both the optical and geometric cross section have units of length squared, the optical efficiency is a unit-less parameter.
While calculating the geometric cross section of spherical particles is a straightforward exercise, determining the geometric cross section of rods, pyramids, and triangular and disc-like plates is less straightforward. There are a number of conventions used. The most common calls for using a cross section of πReff2, where Reff=(3V/4π)(1/3), where V is the total volume of the nanomaterial, i.e. defining the geometric cross to be the cross section of a sphere equal in volume to the nonspherical material.
 Extinction efficiencies of silver nanospheres as a function of nanosphere diameter.
Extinction efficiencies of silver nanospheres as a function of nanosphere diameter.