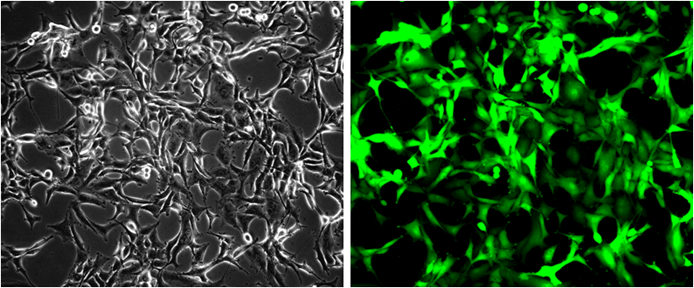
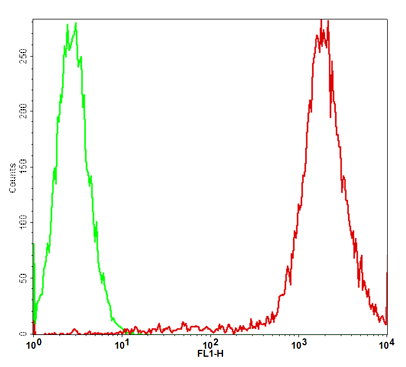
GFP/HeLa Stable Cell Line
Catalogue Number: 14-904ACL-ABO
| Manufacturer: | Abeomics |
| Type: | Cell Lines |
| Shipping Condition: | Blue Ice |
| Storage Condition: | Liquid N2 |
| Unit(s): | 1 vial |
| Application: | FACS, FA |
Description
Description: GFP/HeLa Stable Cell Line is a stably transfected HeLa cell line which expresses enhanced green fluorescent protein (eGFP). Sequence data: Amino acid sequence of eGFP MVSKGEELFTGVVPILVELDGDVNGHKFSVSGEGEGDATYGKLTLKFIC TTGKLPVPWPTLVTTLTYGVQCFSRYPDHMKQHDFFKSAMPEGYVQE RTIFFKDDGNYKTRAEVKFEGDTLVNRIELKGIDFKEDGNILGHKLEYNY NSHNVYIMADKQKNGIKVNFKIRHNIEDGSVQLADHYQQNTPIGDGP VLLPDNHYLSTQSALSKDPNEKRDHMVLLEFVTAAGITLGMDELYK
Additional Text
Storage Note
Immediately upon receipt, store in liquid nitrogen.
Application Notes
Application:. Screen for GFP through Flow Cytometry. Screen for GFP through Fluorescence Microscopy. Culture conditions: Cells should be grown at 37oC with 5% CO2 using DMEM medium (w/ L-Glutamine, 4.5g/L Glucose and Sodium Pyruvate) supplemented with 10% heat-inactivated FBS and 1% Pen/Strep, plus 3 µg/ml of Puromycin. It is recommended to quickly thaw the frozen cells upon receipt or from liquid nitrogen in a 37oC water-bath, transfer to a tube containing 10 ml of growth medium without Puromycin, spin down cells, resuspend cells in pre-warmed growth medium without Puromycin, transfer resuspended cells to T25 flask and culture in 37oC-CO2 incubator. Leave the T25 flask in the incubator for 1~2 days without disturbing or changing the medium until cells completely recover viability and become adherent. Once cells are over 90% adherent, remove growth medium and passage the cells through trypsinization and centrifugation. At first passage, switch to growth medium containing Puromycin. Cells should be split before they reach complete confluence. To passage the cells, detach cells from culture vessel with Trypsin/EDTA, add complete growth medium and transfer to a tube, spin down cells, resuspend cells and seed appropriate aliquots of cells suspension into new culture vessels. Subcultivation ration = 1:10 to 1:20 weekly. To achieve satisfactory results, cells should not be passaged over 16 times.