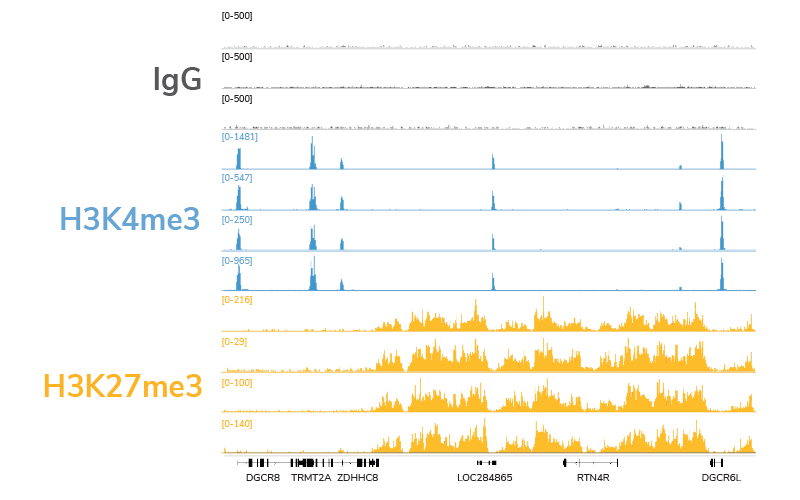
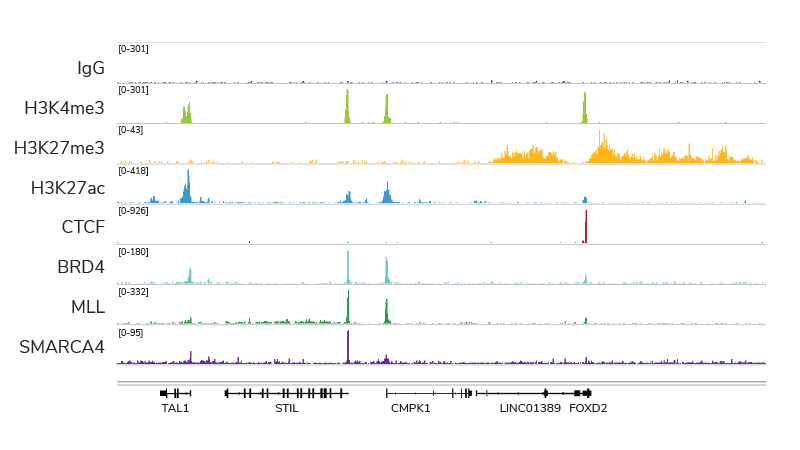
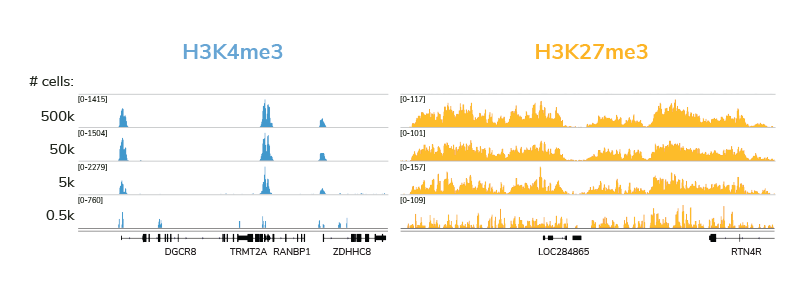
CUT&RUN is quickly becoming a leading assay for chromatin profiling, and it’s easy to see why. The targeted cleavage of antibody-bound chromatin allows for streamlined sample processing and greater signal : noise, meaning researchers can generate high quality data with fewer cells and sequencing reads. EpiCypher’s CUTANA™ ChIC / CUT&RUN Kit comes with nearly everything you will need for a successful CUT&RUN experiment, and each reagent has been meticulously validated and re-validated, from the pAG-MNase enzyme down to the included DNA purification columns.
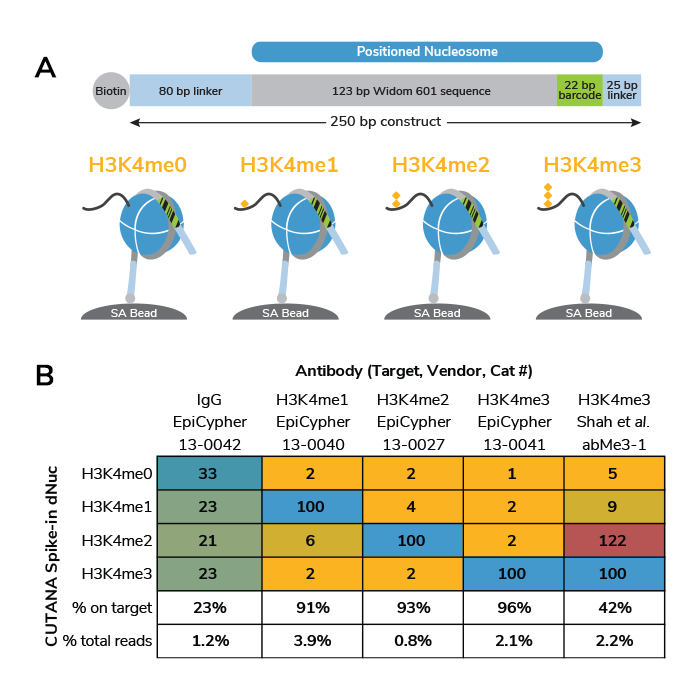
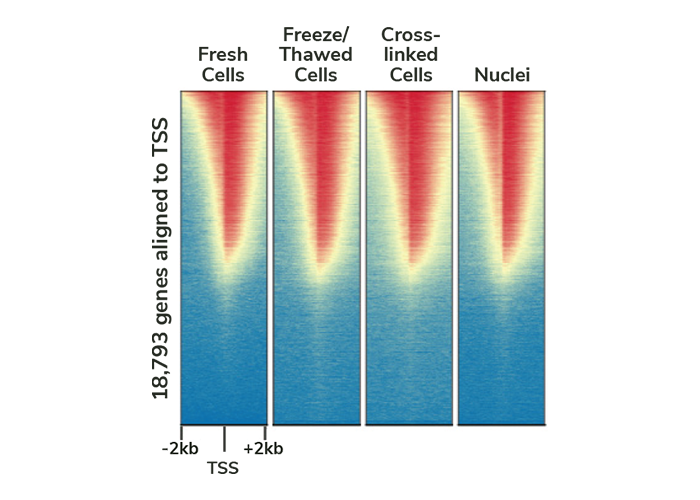
Our thorough kit optimization also resulted in the development of a robust CUT&RUN protocol, validated for diverse targets, sample types, as well as low cell inputs (see below). Furthermore, the CUTANA CUT&RUN Kit comes with all the necessary experimental controls, including positive and negative control antibodies, E. coli spike-in DNA for data normalization, and a panel of DNA-barcoded recombinant modified designer nucleosomes (dNucs™) to monitor the success of your CUT&RUN experiment. Combined, these features make our CUTANA CUT&RUN Kit a standout tool for accurate and user-friendly chromatin profiling.