Dot Blot: A Quick and Easy Method for Separation-Free Protein Detection

How does dot blot work?
Dot blots are like Western blots in that they involve antibody-based detection of membrane-bound proteins. However, the main difference between the two techniques is that dot blotting does not require electrophoretic separation. During a dot blot, the sample (typically a cell or tissue lysate, or a recombinant protein) is spotted directly onto a nitrocellulose or PVDF membrane, which is then blocked prior to incubation with the primary antibody. After washing to remove any unbound antibody reagent, a secondary antibody labeled with a detection moiety such as horseradish peroxidase (HRP) or a fluorescent dye is added to enable target detection.
What is dot blot used for?
Dot blot has several uses. First, it offers a quick and easy method for checking whether a sample contains a particular protein. Provided the primary antibody is specific to the target of interest, dot blot yields rapid visual confirmation of its presence.
Second, dot blot allows researchers to estimate the target protein concentration in a sample. This involves spotting known concentrations of a control (e.g., a recombinant protein) onto the membrane, against which the sample can be compared.
Third, dot blot is used for confirming whether an antibody exhibits specific binding, a strategy that is commonly employed for secondary antibody validation. Figure 1 shows a dot blot verifying the specificity of five HRP-conjugated secondary antibodies raised in goat against different murine subclasses.
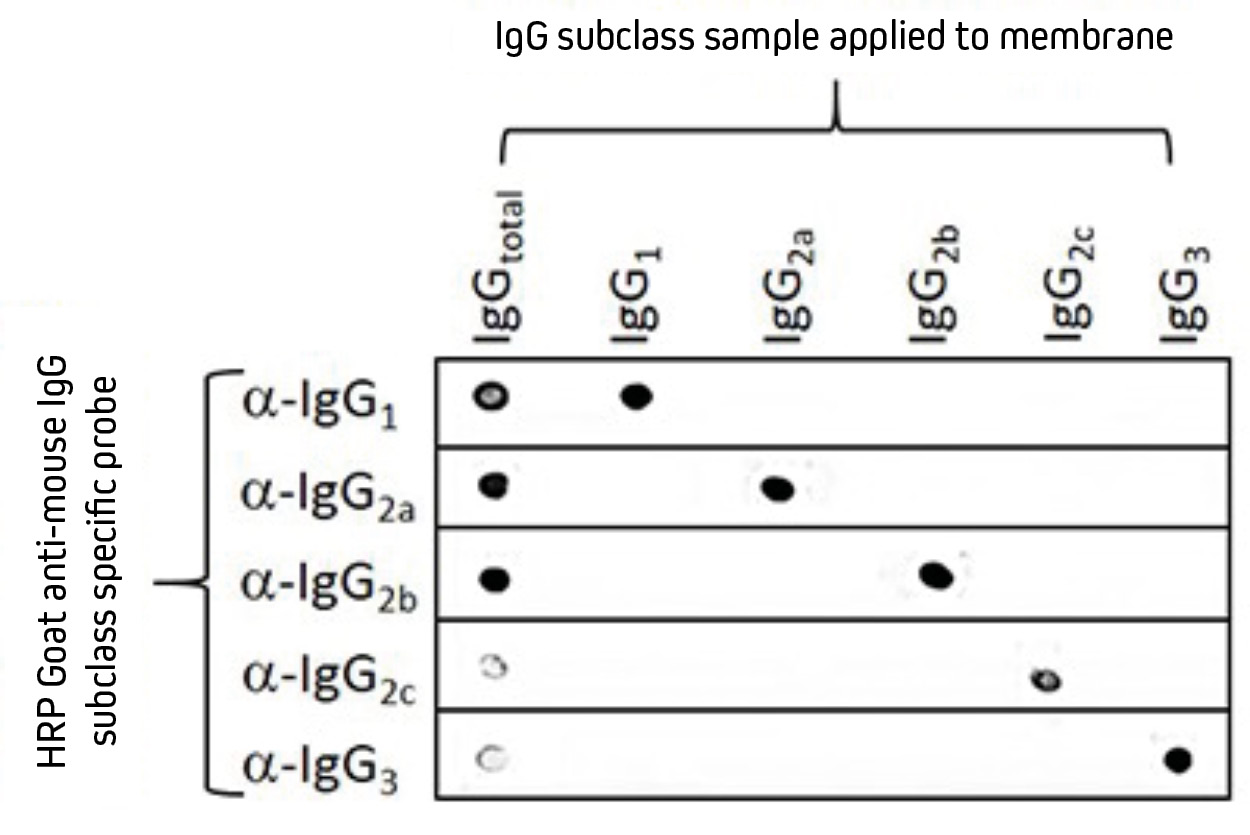
Figure 1. Dot blot showing the specificity of HRP-conjugated goat anti-mouse IgG, Fcγ subclass specific antibodies.
Separate nitrocellulose strips (rows) received 100 ng “dots” of mouse IgG and each subclass, then were blocked with 5% (w/v) BSA in PBST. After probing with HRP-conjugated goat anti-mouse subclass specific antibodies – IgG1 (115-035-205), IgG2a (115-035-206), IgG2b (115-035-207), IgG2c (115-035-208), and IgG3 (115-035-209) – the strips were developed with TMBM substrate. The grid of positive signals shows the specificity of each subclass directed antibody. Some subclasses (α-IgG2c and α-IgG3) are poorly represented in a total IgG pool (IgGtotal) and thus give weak signal.
Finally, dot blot is widely used for optimizing the concentrations of primary and secondary antibodies that are intended for Western blotting. In this scenario, the primary antibody is spotted directly on to the membrane before being detected with the secondary antibody. Often, the dot blot is configured in a grid pattern to simplify the identification of the optimal primary: secondary concentration pair. This can be seen in Figure 2.

Figure 2. Grid-based optimization of primary and secondary antibody concentrations.
ChromPure Rabbit IgG (H+L) was spotted directly onto the membrane for detection with different concentrations of Alexa Fluor® 488 Goat Anti-Rabbit IgG (H+L).
What are the advantages and disadvantages of dot blot?
A major advantage of dot blot over Western blot is that it is much faster. Dot blot is also more cost-effective, since it eliminates the need for materials such as polyacrylamide gels, molecular weight markers, and all of the equipment and buffers required for gel running and protein transfer. In addition, dot blot enables screening of large numbers of samples for the presence of a protein of interest, without being limited by the number of lanes available on a gel.
On the flip side, dot blot has several drawbacks resulting from the lack of protein separation. These include the inability to determine the molecular weight, isoform, or integrity of a protein, and the potential for higher background. Dot blot also precludes monitoring the expression of a normal and modified target within the same blot. Moreover, because dot blot does not generally include a method for normalization (e.g., detection of a housekeeping protein), accurately comparing protein levels between different samples is not possible.
Considerations for dot blot
When performing a dot blot, several important factors should be considered. Knowing whether your primary antibody recognizes native or denatured protein is critical, since this will dictate the choice of dilution buffer. It is also important that the dot blot is oriented correctly to avoid drawing incorrect conclusions; making a small pencil mark on one corner of the membrane can be useful here. When manually spotting proteins (rather than using a commercial dot blot apparatus), smaller volumes should be applied to the membrane to account for sample diffusion. Finally, controls are essential to ensure results are correctly interpreted. Depending on the aim of the experiment, these might be samples known to lack, or exhibit high expression, of the target of interest, or could comprise a ‘no primary antibody’ blot to check for non-specific secondary antibody binding to the sample.
