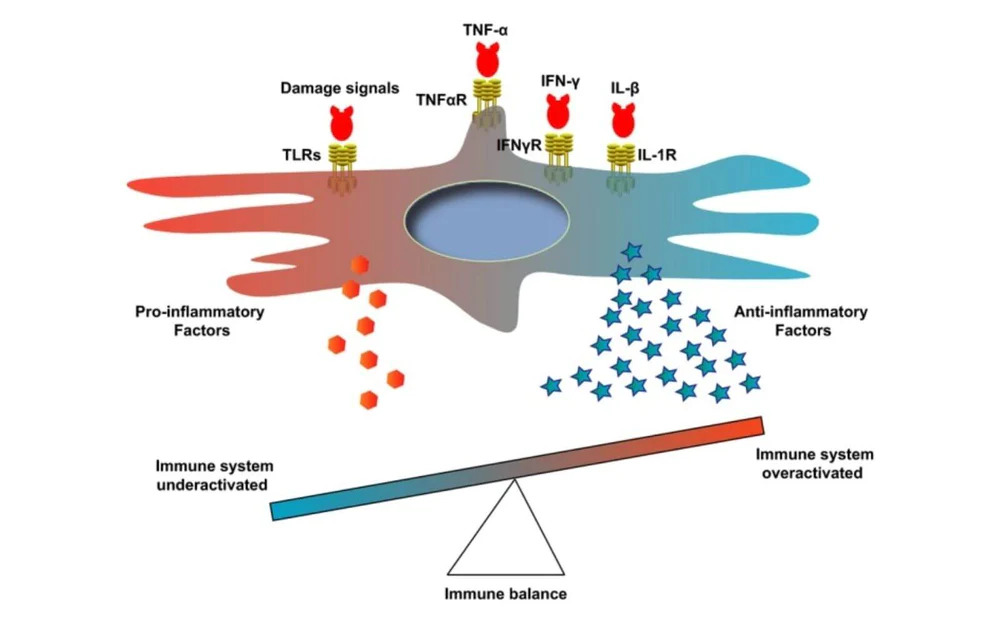
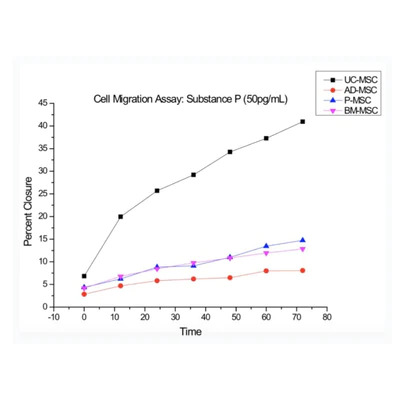
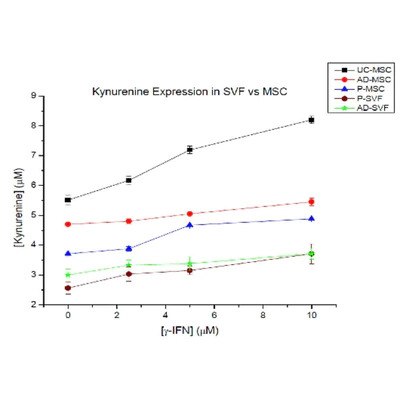
At Vitro Biopharma, our goal is to become a leader in developing differentiated product candidates in a rapid and efficient manner for the immunology and inflammatory market, and ultimately, to address well-established multi-billion-dollar commercial markets. We are committed to translating innovative science into medicines that transform the lives of patients with various diseases.
We believe our 30 years of drug discovery and development has enabled us to identify and advance multiple product candidates from preclinical studies into clinical trials. Our extensive knowledge of immunology and inflammatory conditions informs our decision making to advance the scientific and clinical path to demonstrate proof-of-concept, with the goal of achieving an efficient timeframe and cost-effective budget. The current infrastructure within our discovery and development capabilities of Institutional Review Board (IRB) approved clinical trials in international countries, has allowed us to refine our processes and protocols, as well as, see evidence of efficacy and safety.