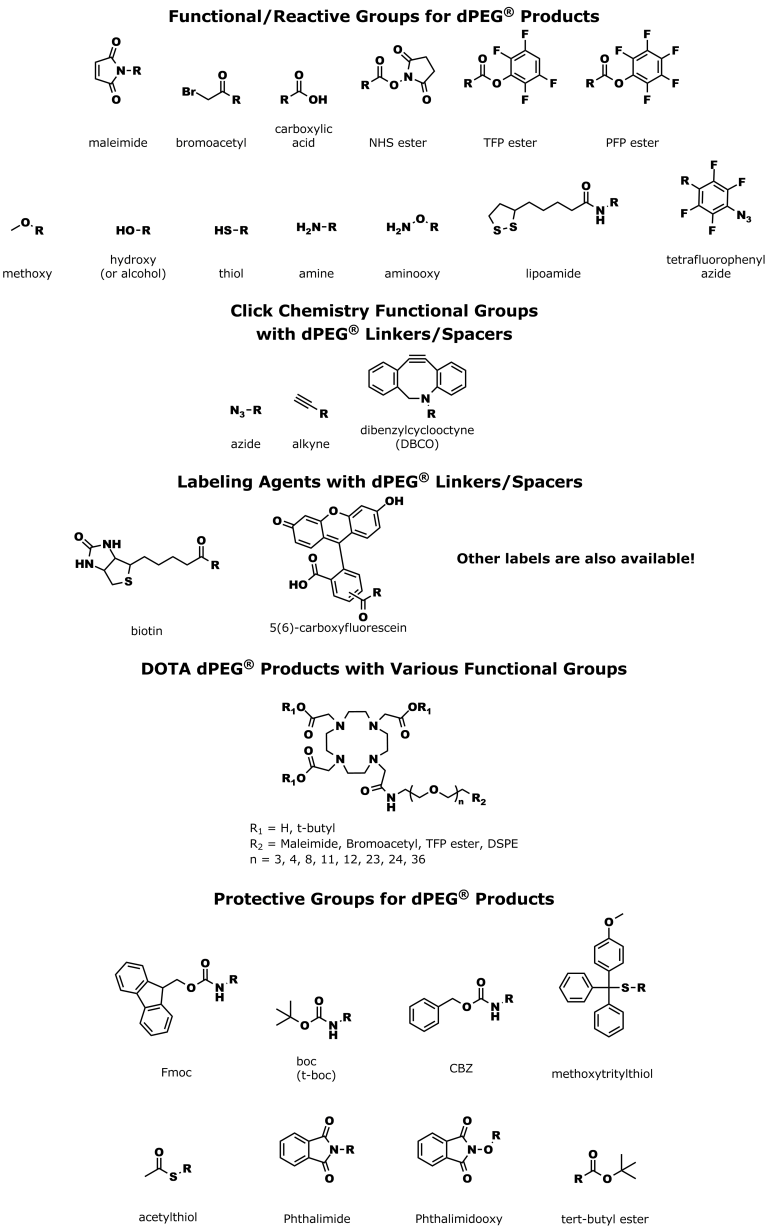
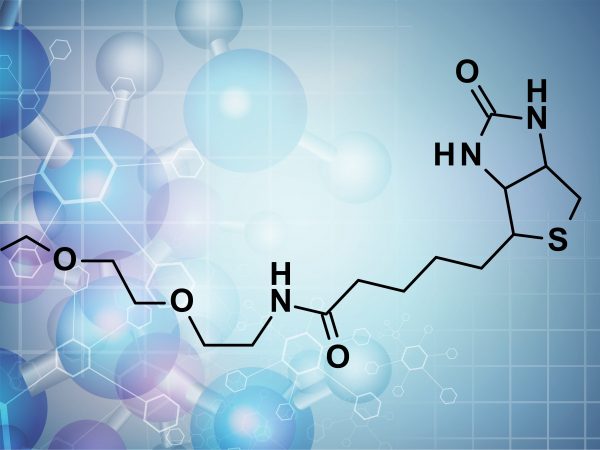
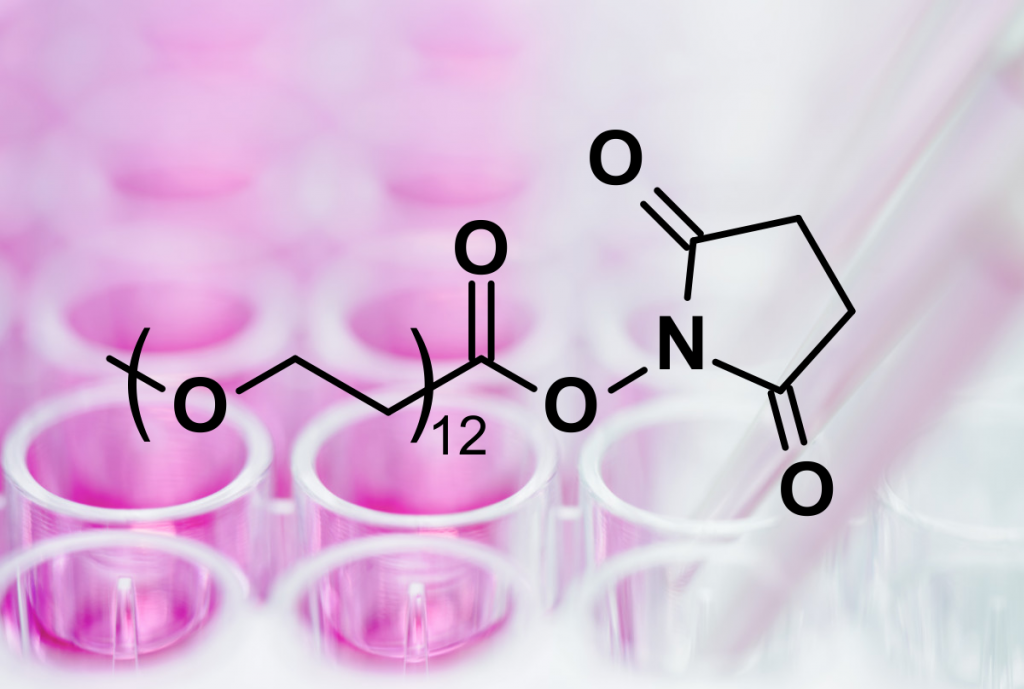
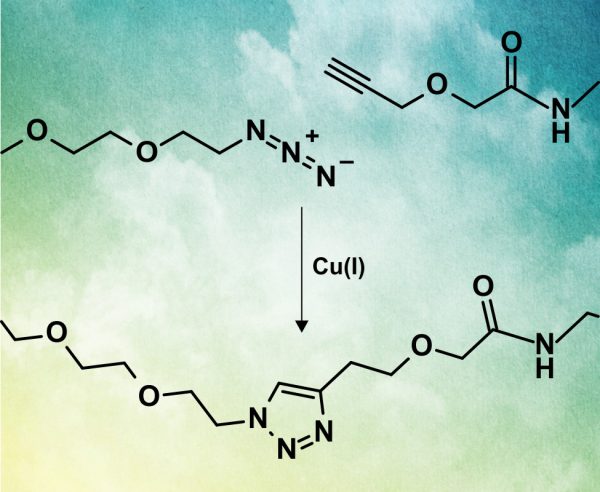
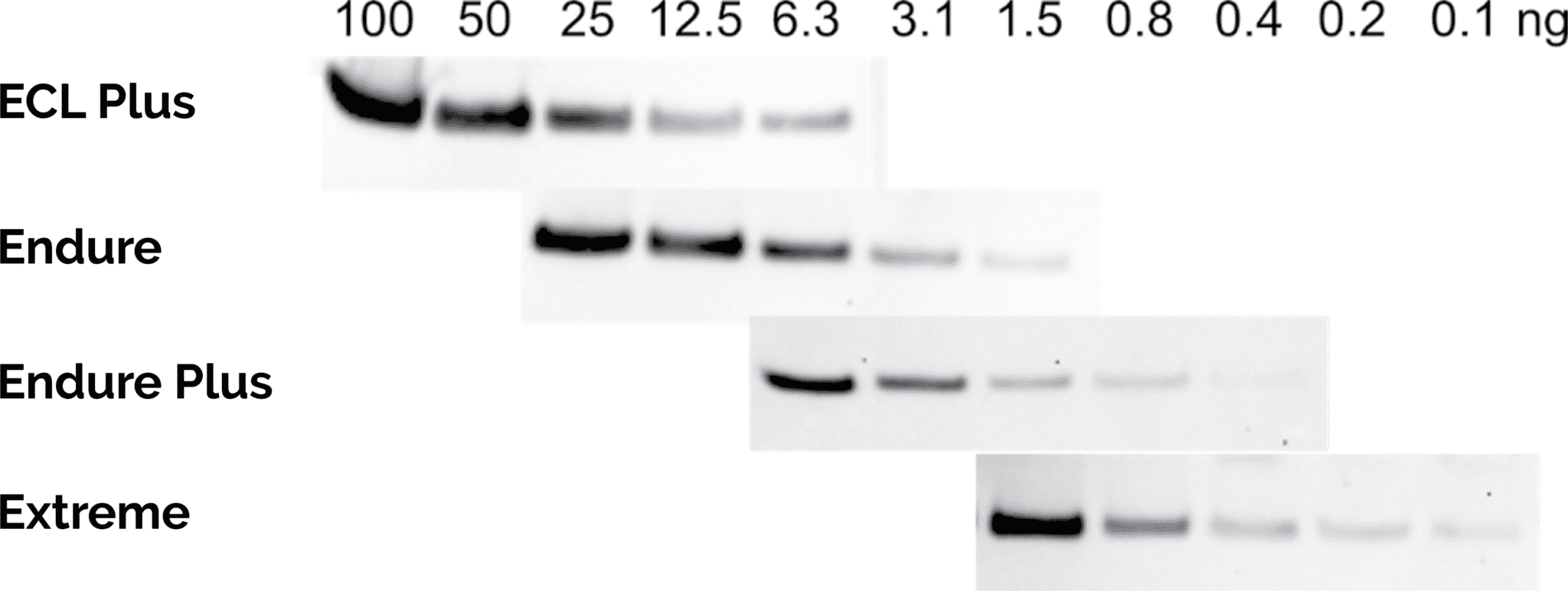
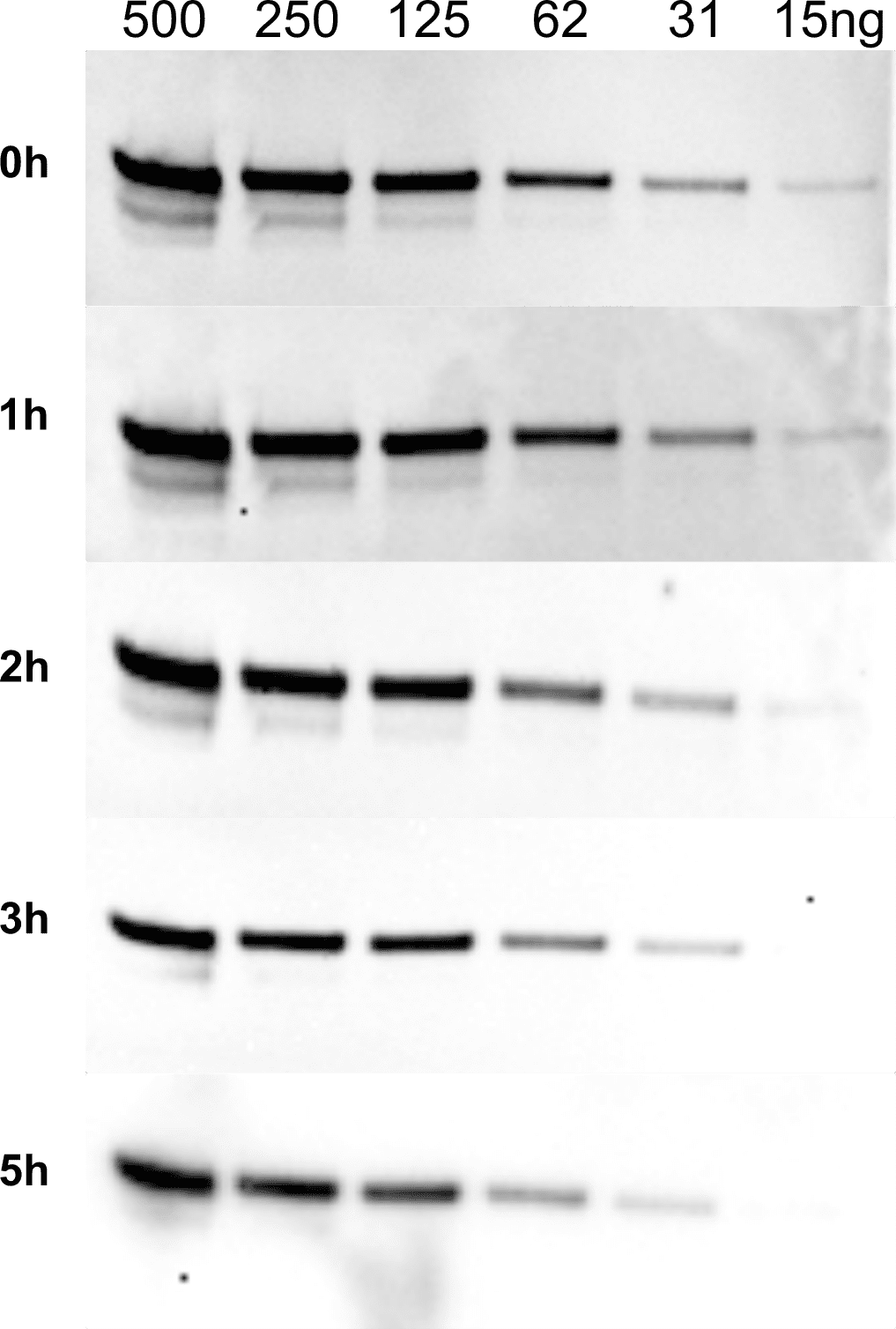
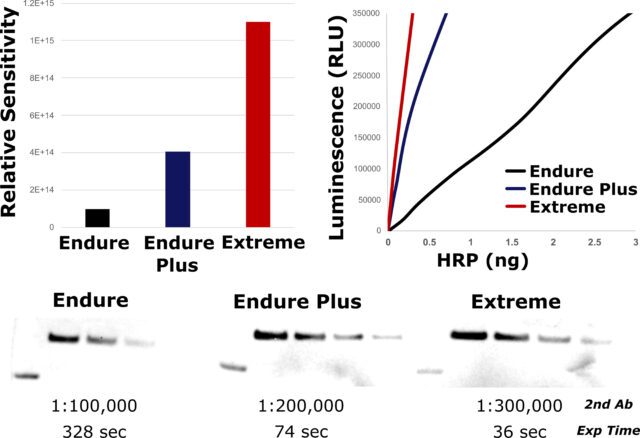
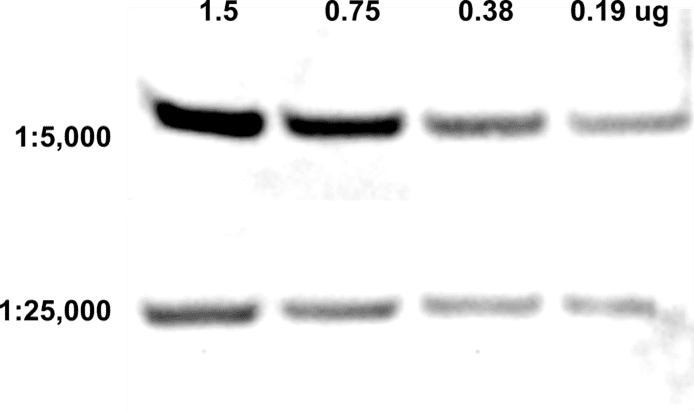
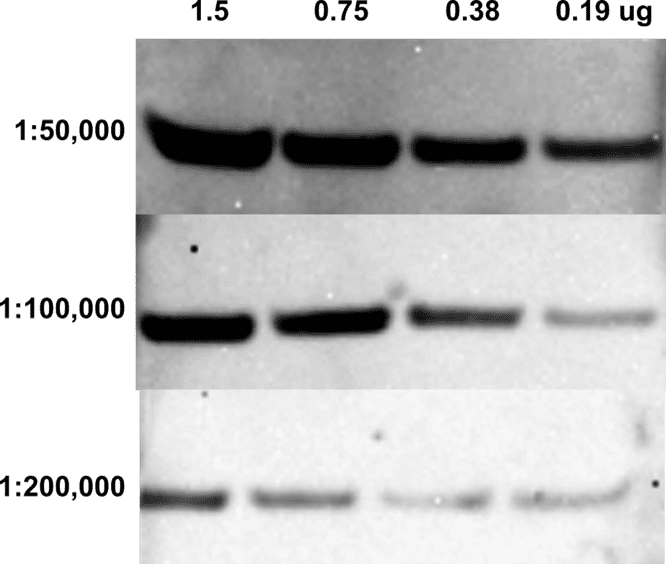
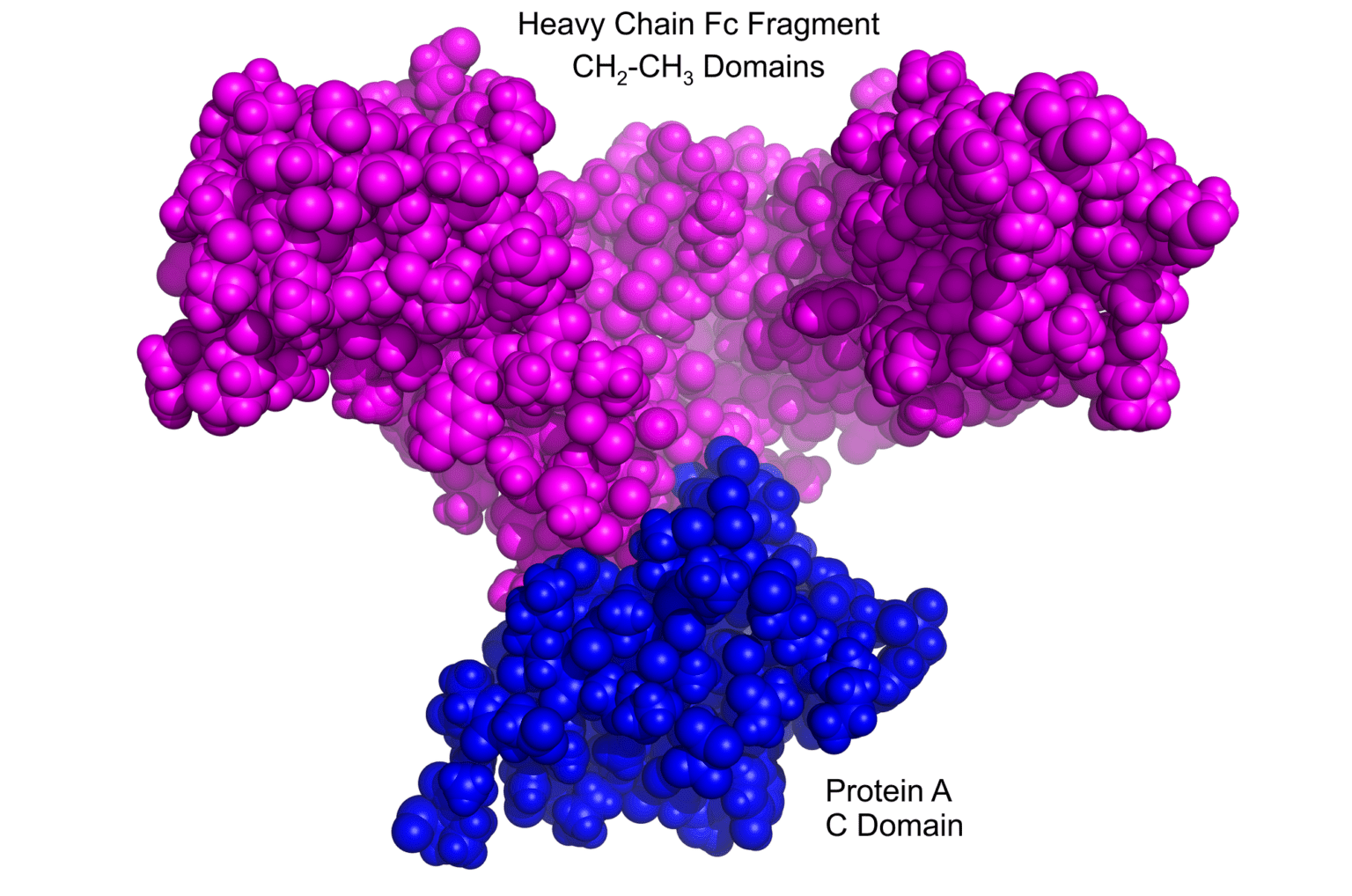
| Product | Name |
| AQ-30151 | Goat anti-Chicken IgY (H&L), AQuora® 550 |
| AQ-30163 | Goat anti-Chicken IgY (H&L), AQuora® 550 min X Mouse, Rabbit, Human, Bovine, Horse |
| AQ-30152 | Goat anti-Chicken IgY (H&L), AQuora® 650 |
| AQ-30164 | Goat anti-Chicken IgY (H&L), AQuora® 650 min X Mouse, Rabbit, Human, Bovine, Horse |
| AQ-30153 | Goat anti-Chicken IgY (H&L), AQuora® 750 |
| AQ-30165 | Goat anti-Chicken IgY (H&L), AQuora® 750 min X Mouse, Rabbit, Human, Bovine, Horse |
| AQ-30154 | Goat anti-Chicken IgY (H&L), AQuora® 800 |
| AQ-30166 | Goat anti-Chicken IgY (H&L), AQuora® 800 min X Mouse, Rabbit, Human, Bovine, Horse |
| AQ-30150 | Goat anti-Chicken IgY (H&L), AQuora® Fluorescein |
| AQ-30162 | Goat anti-Chicken IgY (H&L), AQuora® Fluorescein min X Mouse, Rabbit, Human, Bovine, Horse |
| AQ-30155 | Goat anti-Chicken IgY (H&L), AQuora® TX595 |
| AQ-30167 | Goat anti-Chicken IgY (H&L), AQuora® TX595 min X Mouse, Rabbit, Human, Bovine, Horse |
| AQ-30103 | Goat anti-Human IgA, α chain specific, AQuora® 550 |
| AQ-30104 | Goat anti-Human IgA, α chain specific, AQuora® 650 |
| AQ-30105 | Goat anti-Human IgA, α chain specific, AQuora® 750 |
| AQ-30106 | Goat anti-Human IgA, α chain specific, AQuora® 800 |
| AQ-30102 | Goat anti-Human IgA, α chain specific, AQuora® Fluorescein |
| AQ-30107 | Goat anti-Human IgA, α chain specific, AQuora® TX595 |
| AQ-30091 | Goat anti-Human IgG (Fc), AQuora® 550 |
| AQ-30092 | Goat anti-Human IgG (Fc), AQuora® 650 |
| AQ-30093 | Goat anti-Human IgG (Fc), AQuora® 750 |
| AQ-30094 | Goat anti-Human IgG (Fc), AQuora® 800 |
| AQ-30090 | Goat anti-Human IgG (Fc), AQuora® Fluorescein |
| AQ-30095 | Goat anti-Human IgG (Fc), AQuora® TX595 |
| AQ-30067 | Goat anti-Human IgG (H&L), AQuora® 550 |
| AQ-30068 | Goat anti-Human IgG (H&L), AQuora® 650 |
| AQ-30069 | Goat anti-Human IgG (H&L), AQuora® 750 |
| AQ-30070 | Goat anti-Human IgG (H&L), AQuora® 800 |
| AQ-30066 | Goat anti-Human IgG (H&L), AQuora® Fluorescein |
| AQ-30071 | Goat anti-Human IgG (H&L), AQuora® TX595 |
| AQ-30115 | Goat anti-Human IgM, Fc5µ fragment specific, AQuora® 550 |
| AQ-30116 | Goat anti-Human IgM, Fc5µ fragment specific, AQuora® 650 |
| AQ-30117 | Goat anti-Human IgM, Fc5µ fragment specific, AQuora® 750 |
| AQ-30118 | Goat anti-Human IgM, Fc5µ fragment specific, AQuora® 800 |
| AQ-30114 | Goat anti-Human IgM, Fc5µ fragment specific, AQuora® Fluorescein |
| AQ-30119 | Goat anti-Human IgM, Fc5µ fragment specific, AQuora® TX595 |
| AQ-30171 | Goat anti-Mouse IgG (H&L), AQuora® 488 min X Rabbit, Human, Bovine, Horse, Swine |
| AQ-30017 | Goat anti-Mouse IgG (H&L), AQuora® 550 |
| AQ-30029 | Goat anti-Mouse IgG (H&L), AQuora® 550 min X Rabbit, Human, Bovine, Horse |
| AQ-30018 | Goat anti-Mouse IgG (H&L), AQuora® 650 |
| AQ-30030 | Goat anti-Mouse IgG (H&L), AQuora® 650 min X Rabbit, Human, Bovine, Horse |
| AQ-30019 | Goat anti-Mouse IgG (H&L), AQuora® 750 |
| AQ-30031 | Goat anti-Mouse IgG (H&L), AQuora® 750 min X Rabbit, Human, Bovine, Horse |
| AQ-30020 | Goat anti-Mouse IgG (H&L), AQuora® 800 |
| AQ-30032 | Goat anti-Mouse IgG (H&L), AQuora® 800 min X Rabbit, Human, Bovine, Horse |
| AQ-30027 | Goat anti-Mouse IgG (H&L), AQuora® APC min X Rabbit, Human, Bovine, Horse |
| AQ-30016 | Goat anti-Mouse IgG (H&L), AQuora® Fluorescein |
| AQ-30028 | Goat anti-Mouse IgG (H&L), AQuora® Fluorescein min X Rabbit, Human, Bovine, Horse |
| AQ-30026 | Goat anti-Mouse IgG (H&L), AQuora® PE min X Rabbit, Human, Bovine, Horse |
| AQ-30021 | Goat anti-Mouse IgG (H&L), AQuora® TX595 |
| AQ-30033 | Goat anti-Mouse IgG (H&L), AQuora® TX595 min X Rabbit, Human, Bovine, Horse |
| AQ-30034 | Goat anti-Mouse IgG (H&L), PE HS min X Rabbit, Human, Bovine, Horse |
| AQ-30043 | Goat anti-Rabbit IgG (H&L), AQuora® 650 |
| AQ-30046 | Goat anti-Rabbit IgG (H&L), AQuora® TX595 |
| AQ-30042 | Goat anti-Rabbit IgG (H&L), AQuora® 550 |
| AQ-30054 | Goat anti-Rabbit IgG (H&L), AQuora® 550 min X Mouse, Human, Bovine, Horse |
| AQ-30055 | Goat anti-Rabbit IgG (H&L), AQuora® 650 min X Mouse, Human, Bovine, Horse |
| AQ-30044 | Goat anti-Rabbit IgG (H&L), AQuora® 750 |
| AQ-30056 | Goat anti-Rabbit IgG (H&L), AQuora® 750 min X Mouse, Human, Bovine, Horse |
| AQ-30045 | Goat anti-Rabbit IgG (H&L), AQuora® 800 |
| AQ-30057 | Goat anti-Rabbit IgG (H&L), AQuora® 800 min X Mouse, Human, Bovine, Horse |
| AQ-30052 | Goat anti-Rabbit IgG (H&L), AQuora® APC min X Mouse, Human, Bovine, Horse |
| AQ-30041 | Goat anti-Rabbit IgG (H&L), AQuora® Fluorescein |
| AQ-30053 | Goat anti-Rabbit IgG (H&L), AQuora® Fluorescein min X Mouse, Human, Bovine, Horse |
| AQ-30051 | Goat anti-Rabbit IgG (H&L), AQuora® PE min X Mouse, Human, Bovine, Horse |
| AQ-30058 | Goat anti-Rabbit IgG (H&L), AQuora® TX595 min X Mouse, Human, Bovine, Horse |