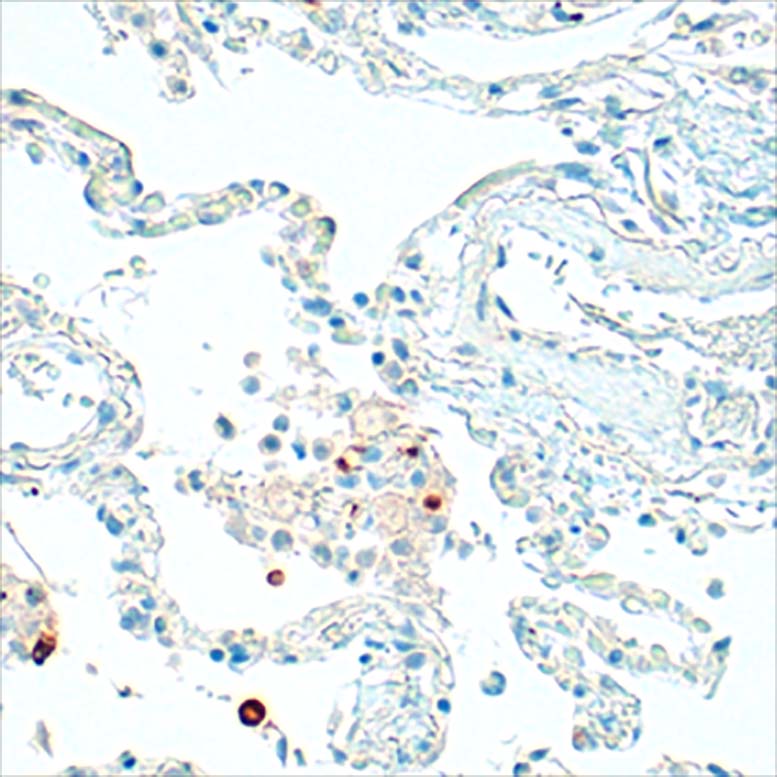
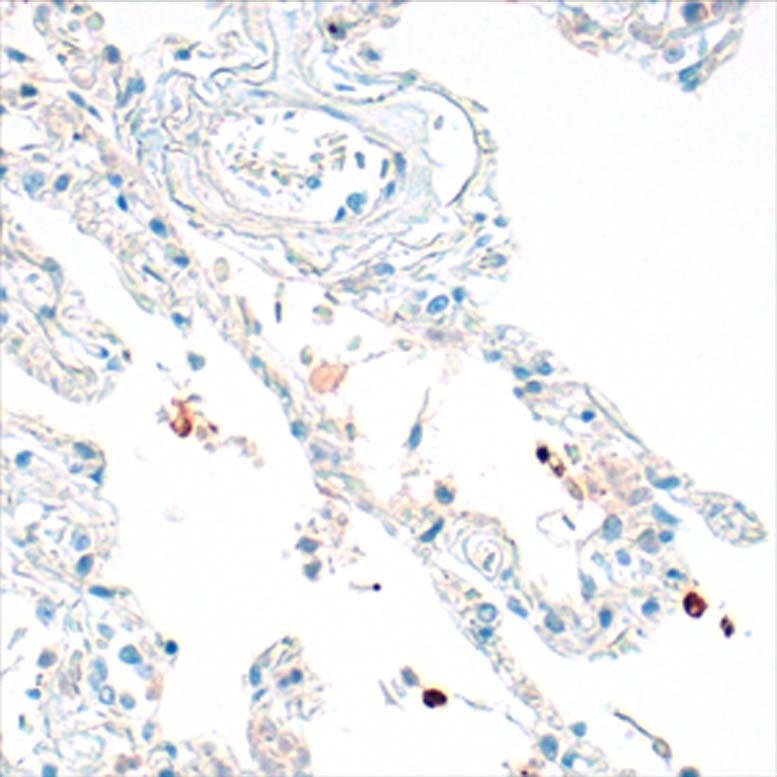
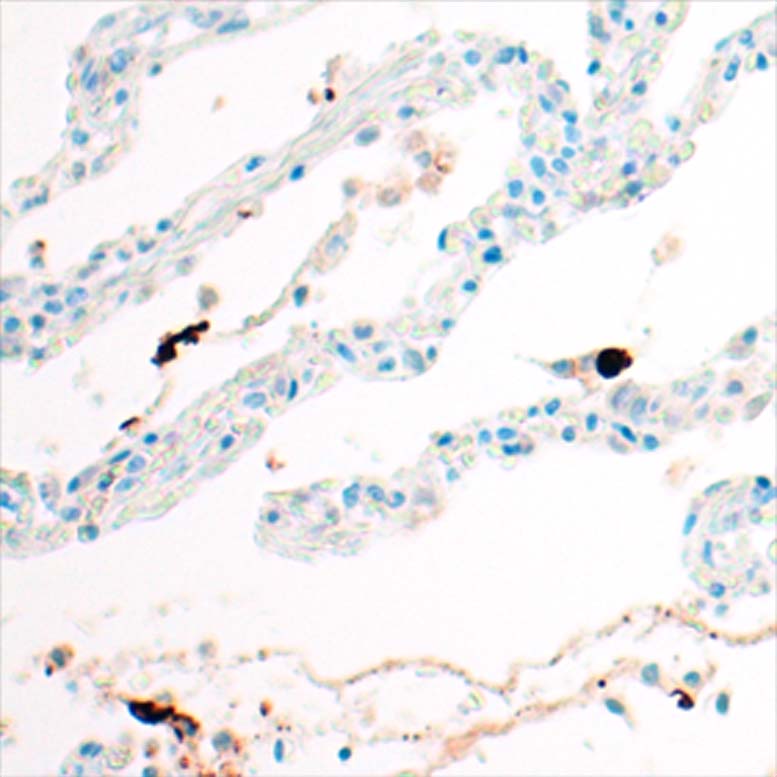
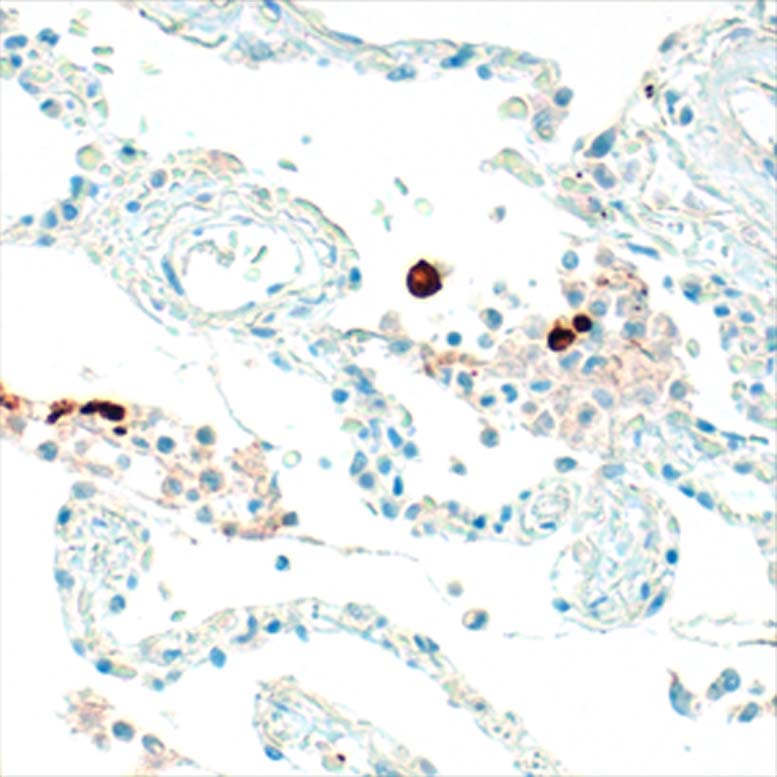
Anti-human IgE antibodies for serolgic analysis and immunoassay design in allergy diagnostics and and allergy research.
Immunoglobulin E (IgE) plays an important role in allergic reactions of the immediate type (type I). The total amount of IgE in blood serum is often elevated in allergic diseases such as allergic rhinitis (including especially hay fever and dust mite allergy) or bronchial asthma, but also in food allergies. Increased total IgE titers in serum can also frequently be found in connection with neurodermatitis (atopic eczema).
Clone GHE20 is already established in various immunoassays of our industry partners for the serolgoic detection of human IgE. From now on, our anti-human IgE (ε) antibody is also available for your serolgic analyses in allergy research or as a component for immunoassays.
We offer numerous anti-human IgE antibody conjugates for different enzymes and fluorescent dyes. As standard variants you receive unconjugated or conjugated antibodies with AP, HRPO, FITC and Biotin. In addition, we can meet your individual requirements and couple almost any conceivable conjugate to the IgE antibody. Use our conjugation service to customize antibodies for your specific test systems.
Use of anti-human IgE antibodies in ELISA
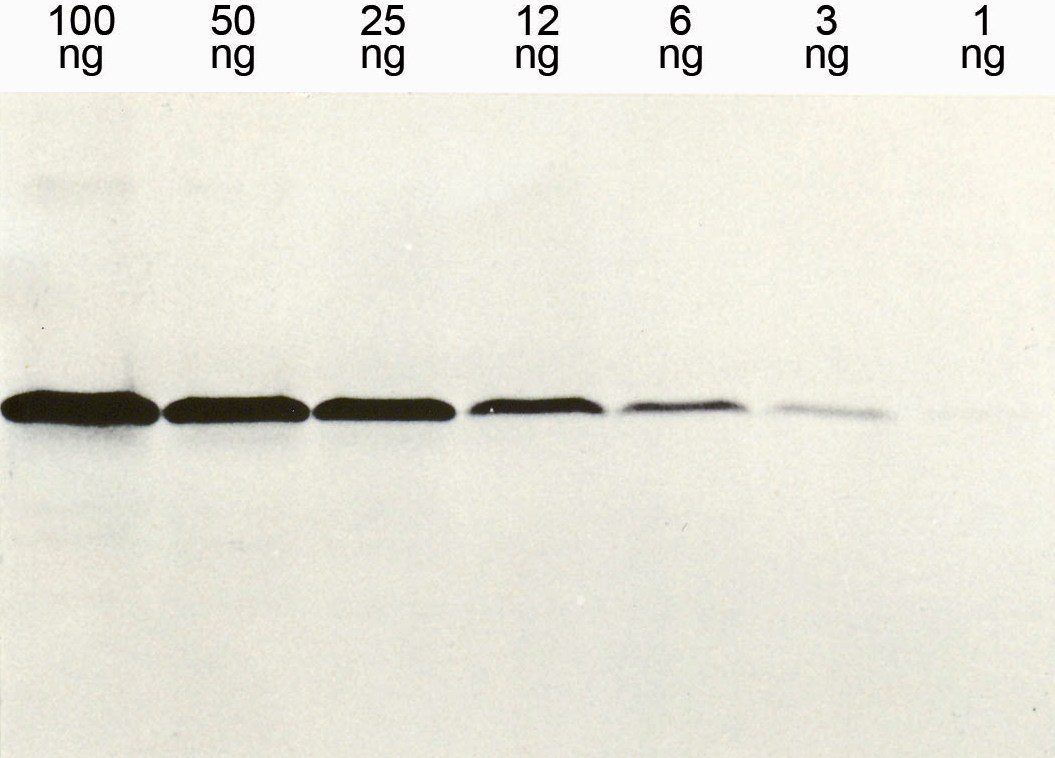
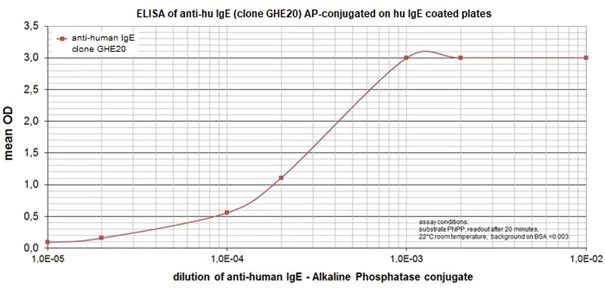
The mouse clone GHE20 is specific for the epsilon (ε)-chain of human IgE and shows no cross-reaction with human IgG, IgM, or IgA. Our anti-human IgE has been tested in ELISA applications and detects human IgE from various sources (e.g. plasma or serum) with high sensitivity.
Figure 1: Illustration of antibody sensitivity in an ELISA with different concentrations of the anti-human IgE clone GHE20 for detection on IgE-coated analysis plates.
| Prod. No. |
Product |
Conjugation |
Qty |
| SCR-180656 |
anti-hu IgE, ms clone GHE20 |
unconj. |
0,1 mg |
| SCR-180657 |
anti-hu IgE, ms clone GHE20 |
unconj. |
0,5 mg |
| SCR-180659 |
anti-hu IgE, ms clone GHE20 |
HRPO |
1 ml |
| SCR-180661 |
anti-hu IgE, ms clone GHE20 |
FITC |
0,5 mg |
| SCR-180663 |
anti-hu IgE, ms clone GHE20 |
Biotin |
0,5 mg |
| SCR-180665 |
anti-hu IgE, ms clone GHE20 |
Alk. Phos. |
1 ml |