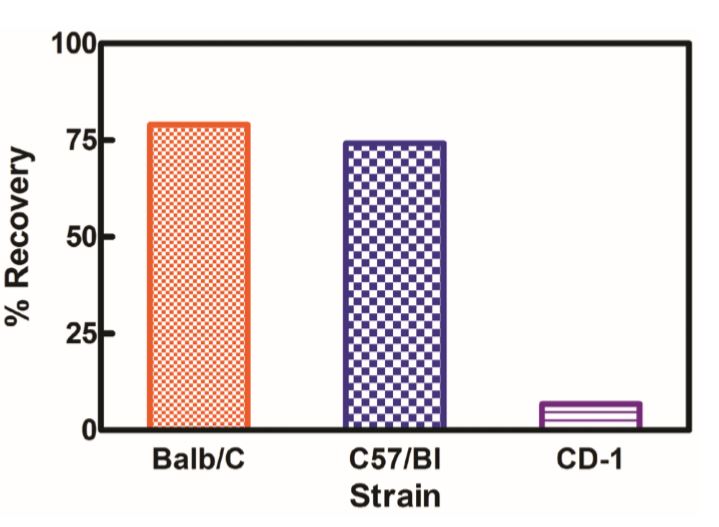
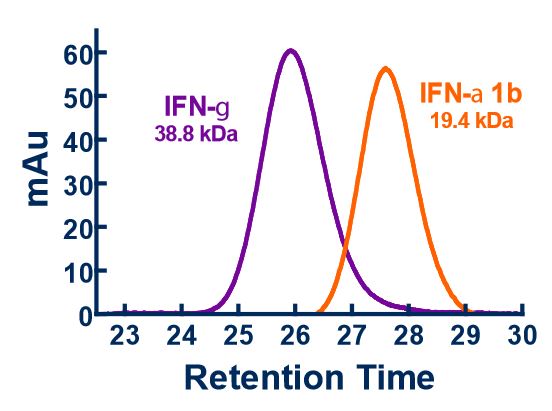
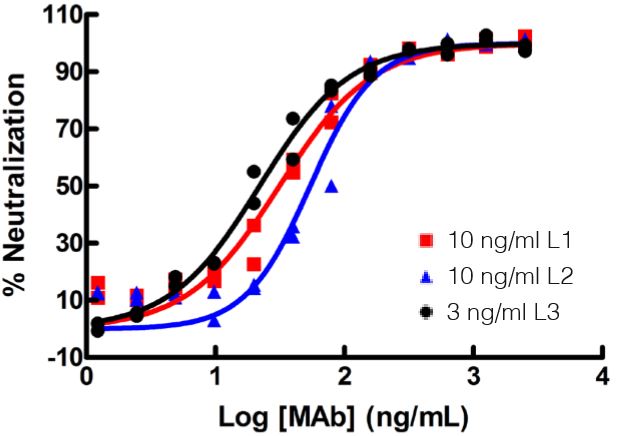
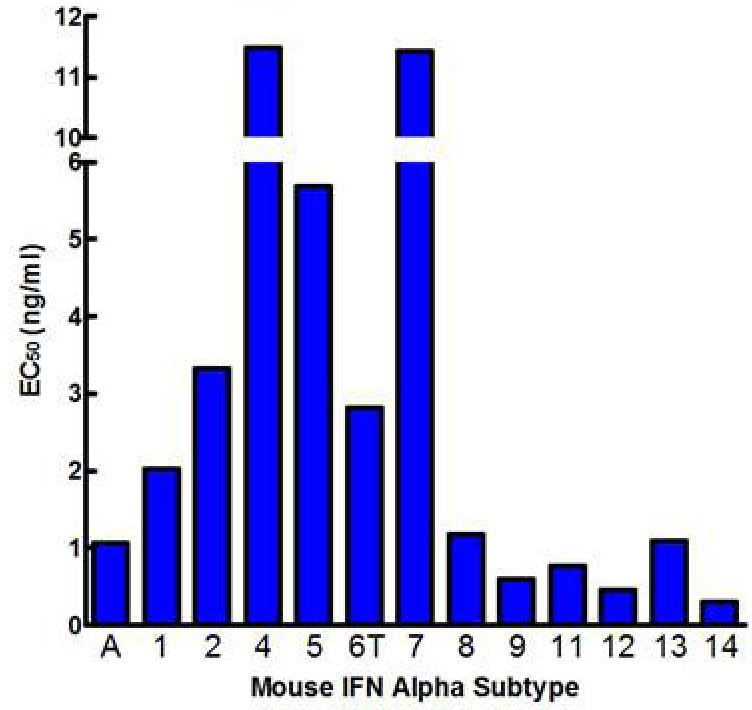
The standard bioassay used to determine the biological activity of interferon (IFN) encompasses measurement of the protection of cells from the cytopathic effect (CPE) of certain viruses. CPE assay services may be used to measure the antiviral activity of IFN-α, β, ω, γ, and λ in human samples, as well as in a variety of other species, and in multiple matrices.
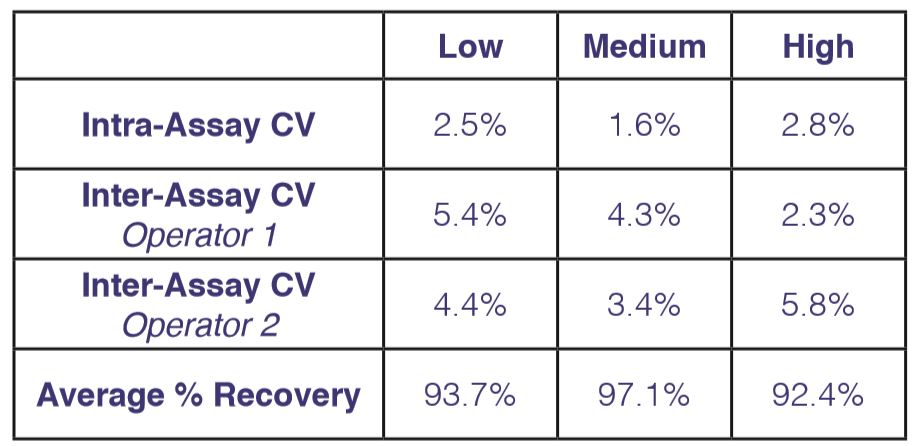
CPE assays are inherently complex due to the metabolic state of cells, virus replication, and the ability of IFN to protect cells. Our years of knowledge and expertise in CPE assays will reduce variability and provide you with reproducible quality results.
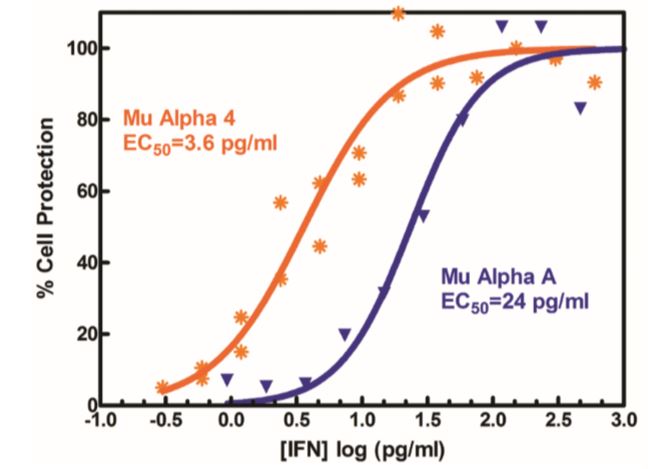
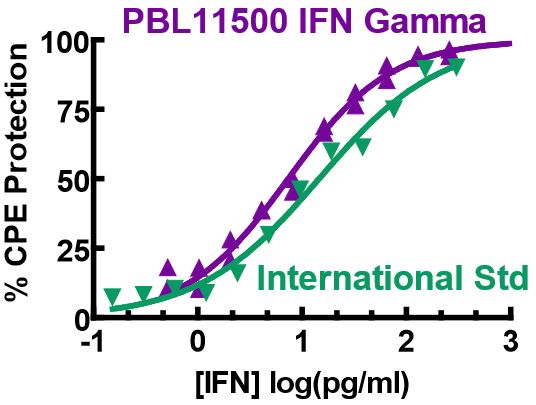
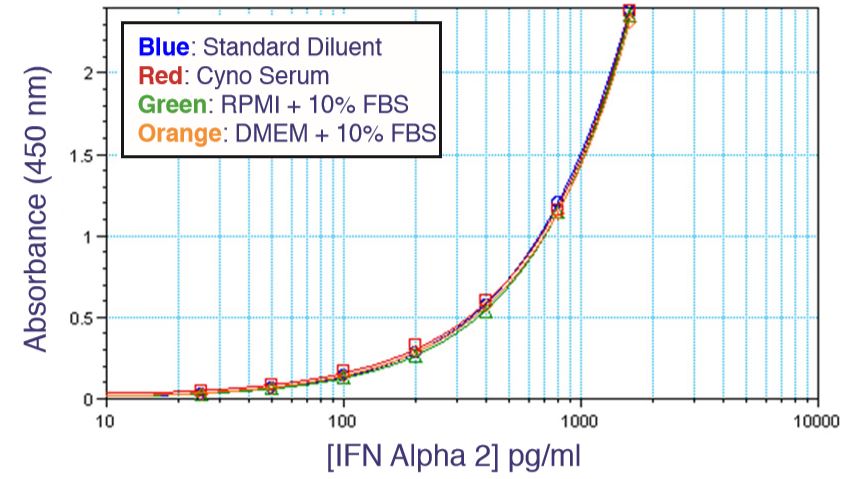
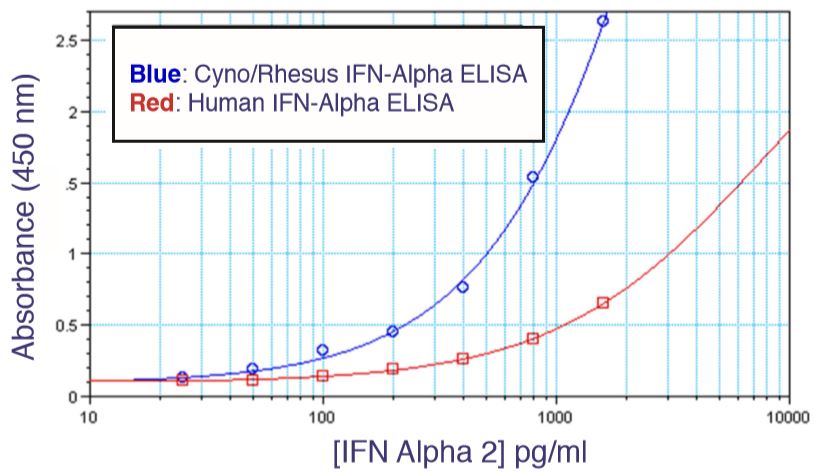
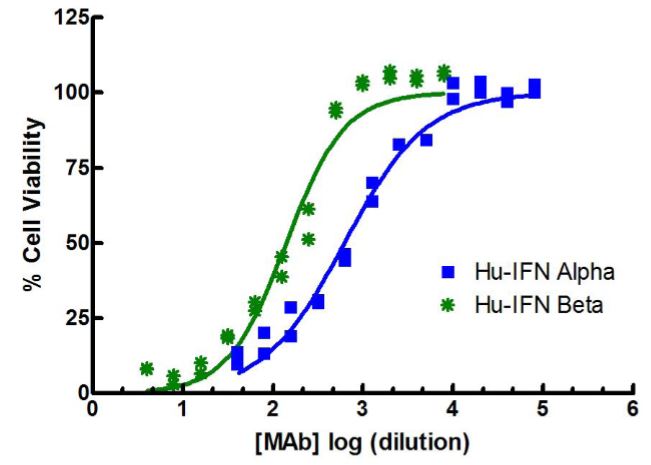
Results are provided either as a graphical representation of dye binding which allows slope and parallel line analysis, or visually determined from microscopic examination of the CPE and determination of the dilution of test samples which protects 50% of the cells.
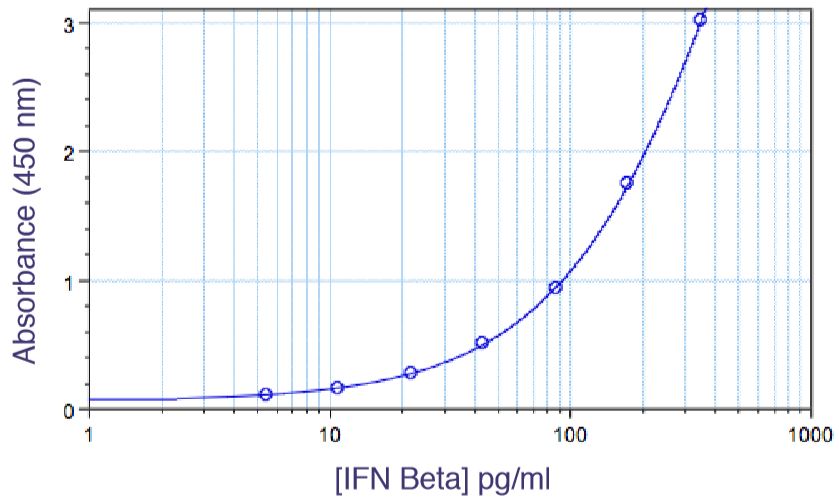
Figure 4 to the right shows IFN-beta titrated in a CPE assay on A549 cells with EMCV. In this antiviral assay, the EC50 is approximately 1 unit/ml of interferon. Units of IFN activity were determined colorimetrically with respect to an international reference standard for human IFN.



.jpg)